antivenom administration for snake bites
- related: toxicology and toxic ingestions
- tags: #literature #icu
Snakebites cause significant morbidity and mortality, especially in the developing world. There are hundreds of species of venomous snakes, with the majority belonging to two families, Viperidae (subfamily Crotalidae) and Elapidae. The Crotalidae are pit vipers that include rattlesnakes, moccasins, and copperheads, and the Elapidae include coral snakes, cobras, and mambas. Crotalidae account for 99% of the envenomations in North America. The identification of a snake species solely based on a victim’s description is unreliable, but the geographic location of the patient when he was bitten suggests that it might have been a water moccasin, a type of pit viper found in the wetlands of the southeastern United States. Pit vipers are capable of controlling the amount of venom released during a bite, depending on whether the bite is defensive or predatory. During a “dry bite,” no venom is released, and a victim may experience no symptoms at all. At the other extreme, a large envenomation has the potential to cause extensive tissue necrosis or even be lethal.
Pit viper antivenom is produced by hyperimmunizing sheep or horses with snake venom. The hyperimmune serum is then purified into specific IgG Fab fragments that bind and neutralize the numerous peptides and proteins that constitute snake venom. Because it has potential side effects, antivenom is reserved for serious envenomations. Guidelines for its use are based on the severity of signs and symptoms. A simple snakebite severity scale is shown in Figure 3.

A score of 2 or more has been advocated as the threshold for antivenom administration, and the World Health Organization also recommends antivenom treatment for patients with a bite on a digit because digits have a higher risk of ischemic necrosis.
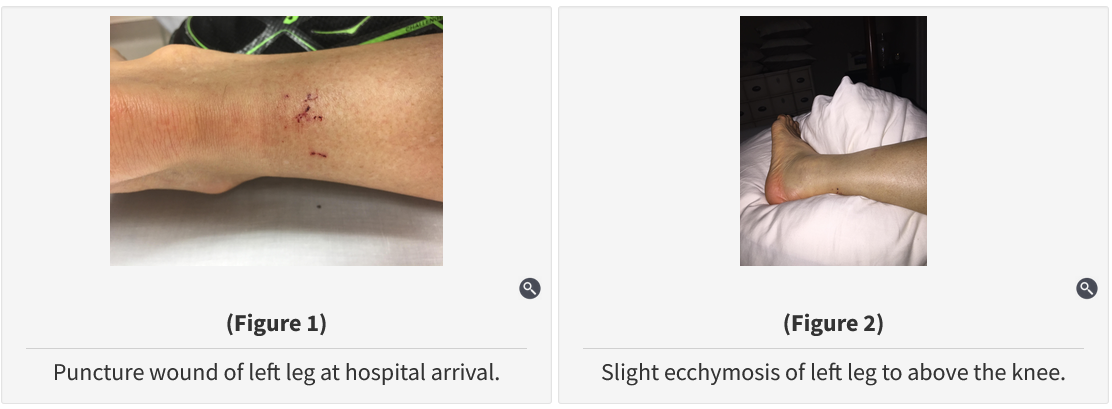
Pit viper envenomation initially causes local injury around the site of the bite. The patient had throbbing pain, swelling, and local ecchymosis, informing us that he had more than a dry bite. His pain and swelling worsened, but he had no evidence of tissue necrosis and did not develop signs of a compartment syndrome. He did, however, develop swelling more than 30 cm from the bite site. In terms of systemic symptoms, he had no evidence of coagulopathy, but he did develop nausea, vomiting, and diarrhea. His score of 3 therefore justifies antivenom treatment.
Crotalidae polyvalent immune Fab (ovine) and Crotalidae immune F(ab)2 (equine) are both approved for use in the United States for pit viper bites and are widely available at referral hospitals. Elapidae antivenom is less widely available but can often be found at zoos and regional poison control centers. The polyvalent antivenom (Crotalidae polyvalent immune Fab) is produced by immunizing animals with venom from multiple snake species. Crotalidae polyvalent immune Fab is preferred; however, monovalent antivenom may still offer cross protection for multiple types of pit vipers. The antivenom is administered intravenously using an algorithm for dosing (see reference section). Potential side effects include fever, pruritus, urticaria, angioedema, anaphylaxis, serum sickness, lymphadenopathy, and proteinuria.
As evidenced by normal pulses and capillary refill, this patient does not have a compartment syndrome at this time, so there is no role for immediate fasciotomy. Ongoing careful monitoring is indicated.
On the basis of several North American studies, the risk of skin and soft-tissue infection after a snakebite is less than 1%, and routine antibiotic prophylaxis is not recommended.12345
Links to this note
Footnotes
-
Appropriate dosing of CroFab achieves initial and sustained control of envenomation. CroFab. Accessed March 18, 2024. https://crofab.com/treating-with-crofab/dosing.html ↩
-
Kim OH, Lee JW, Kim HI, et al. Adverse cardiovascular events after a venomous snakebite in Korea. Yonsei Med J. 2016;57(2):512-517. PubMed ↩
-
Ralph R, Faiz MA, Sharma SK, et al. Managing snakebite. BMJ. 2022;376:e057926. PubMed ↩
-
Russell JJ, Schoenbrunner A, Janis JE. Snake bite management: a scoping review of the literature. Plast Reconstr Surg Glob Open. 2021;9(4):e3506. PubMed ↩