warm autoimmune hemolytic anemia management
- related: Oncology
- tags: #literature #oncology
- transfuse O negative unmatched blood
- avoid fluids
- symptoms
- FiO2 100%: all hemoglobin saturated, use 100% O2
- PaO2 very high (e.g. 500): very saturated blood
This patient has developed warm autoimmune hemolytic anemia, likely associated with CLL. He now presents with severe anemia due to significant hemolysis from warm autoimmune hemolytic anemia. His anemia is life-threatening, as evidenced by his elevated lactate level and marked tachycardia. He needs erythrocytes as soon as possible. His frequent transfusions over the last few months have resulted in the development of numerous antibodies to minor, non-Rh D antigens on erythrocytes. Obtaining matched blood for transfusion from the blood bank will take hours. Given that his severe anemia is resulting in life-threatening hypoxia at the tissue level, he is at risk for severe complications if he waits hours to receive erythrocytes. Severe anemia, defined as a hemoglobin level less than 6 g/dL (60 g/L) or causing hemodynamic compromise from a rapid decrease in hemoglobin is a medical emergency requiring immediate stabilization and usually transfusion. In this situation, the best course of action is to perform transfusion with unmatched O negative (ie, “trauma” or “emergency release”) blood until his condition stabilizes or matched blood is available. If the patient’s blood type is known, type-specific blood can be used instead of unmatched O negative emergency release blood, but only if the type-specific blood can be obtained emergently. A reasonable target for transfusion threshold would be clearing of his serum lactate and resolution of his tachycardia. Studies have suggested that lactic acidosis develops in healthy people around a hemoglobin level of 4 g/dL (40 g/L). Once his condition has stabilized, specific warm autoimmune hemolytic anemia treatment can be started, focusing on reducing hemolysis and halting antibody production.
His tachycardia and lactic acidosis are signs of inadequate delivery of oxygen to the tissues. His SpO2 with FIO2 of 1.0 is 100%, indicating that his RBCs are fully saturated with oxygen; he just needs more erythrocytes. With a hemoglobin level of only 2.5 g/dL (25 g/L), his PaO2 of 552 mm Hg contributes significantly to his oxygen-carrying capacity. Patients with severe, critical anemia should receive FIO2 of 1.0 to maximize oxygen-carrying capacity.
Although this patient has lactic acidosis, reflecting inadequate oxygen delivery at the tissue level, it is not secondary to low cardiac output but instead to severe anemia. Administration of IV crystalloids will not increase the patient’s oxygen-carrying capacity or oxygen delivery to the tissues.
Warm autoimmune hemolytic anemia is due to an antibody that is active at normal body temperature. Although warm autoimmune hemolytic anemia can arise spontaneously, most cases arise secondary to a medication or condition, such as CLL, that predisposes the patient to the production of autoantibodies. The autoantibodies are almost always IgG and are typically polyclonal panagglutinins directed against common RBC antigens. Once the patient’s hemodynamic status is stable, first-line treatment for the warm autoimmune hemolytic anemia can be started. First-line therapy is usually glucocorticoids with or without rituximab. Methylprednisolone would treat warm autoimmune hemolytic anemia, but stabilization of the patient’s severe anemia should be performed first. Plasmapheresis is not a primary treatment modality for warm autoimmune hemolytic anemia and is not indicated according to the American Society for Apheresis.1234567
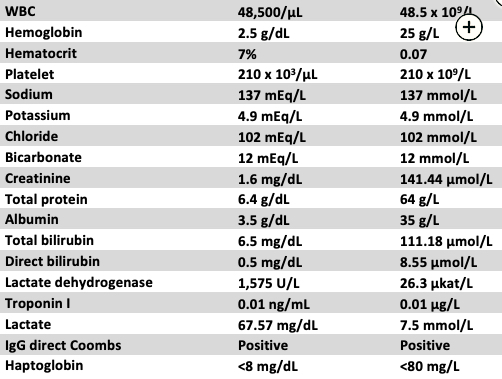
A 57-year-old man with a medical history of hypertension and chronic lymphocytic leukemia (CLL) with transfusion-dependent anemia presents with a 1-day history of progressive shortness of breath and chest pain. For the past 3 months, the patient has received twice weekly blood transfusions for severe refractory anemia secondary to CLL. At presentation, he is in obvious distress, with a respiratory rate of 38/min and supraclavicular retractions, heart rate of 135/min, and BP of 115/64 mm Hg. He is afebrile. He is only able to speak a word or two at a time. His lungs are clear, and his heart is tachycardic but regular with a soft systolic murmur at the apex. His abdominal examination results are notable for splenomegaly. He has no cyanosis or peripheral edema. His SpO2 is 92% breathing room air, and he is using a nonrebreather face mask with FIO2 of 1.0, which increases his SpO2 to 100%. Arterial blood gas measurements with the nonrebreather mask are a pH of 7.30, PCO2 of 30 mm Hg, and PO2 of 552 mm Hg. Other laboratory values are listed in Figure 1. A chest CT scan is remarkable for some mediastinal adenopathy, but the lung fields are clear of infiltrates, and no pulmonary embolism is seen. Blood is sent for type and screen, but the blood bank calls to report that the patient has numerous minor blood antigens and matching blood for transfusion will take a few hours.
What is the best treatment at this time?
Links to this note
Footnotes
-
Berentsen S, Barcellini W. Autoimmune hemolytic anemias. N Engl J Med. 2021;385(15):1407-1419. PubMed ↩
-
Brodsky RA. Warm autoimmune hemolytic anemia. N Engl J Med. 2019;381(7):647-654. PubMed ↩
-
Ness PM. How do I encourage clinicians to transfuse mismatched blood to patients with autoimmune hemolytic anemia in urgent situations? Transfusion. 2006;46(11):1859-1862. PubMed ↩
-
Petz LD. A physician’s guide to transfusion in autoimmune haemolytic anaemia. Br J Haematol. 2004;124(6):712-716. PubMed ↩
-
Salama A, Berghöfer H, Mueller-Eckhardt C. Red blood cell transfusion in warm-type autoimmune haemolytic anaemia. Lancet. 1992;340(8834-8835):1515-1517. PubMed ↩
-
Weiskopf RB, Viele MK, Feiner J, et al. Human cardiovascular and metabolic response to acute, severe isovolemic anemia. JAMA. 1998;279(3):217-221. PubMed ↩