Goodpasture syndrome anti-GBM can develop DAH
- related: DAH diffuse alveolar hemorrhage, goodpasture syndrome anti GBM
- tags: #literature #pulmonology
This patient is presenting with a pulmonary–renal syndrome, and the serology points toward anti-glomerular basement membrane (anti-GBM) antibody disease (also called “Goodpasture syndrome”) as the likely etiology. Treatment consists of corticosteroids, cyclophosphamide, and plasmapheresis. Anti-GBM disease is a renal–pulmonary syndrome causing diffuse alveolar hemorrhage (DAH) and rapidly progressive glomerulonephritis (RPGN) and renal failure. It is a small-vessel vasculitis resulting from circulating autoantibodies against an antigen component of the basement membrane of the glomerulus and alveoli. It is relatively rare and is seen in young adults usually <30 years old, with a slight male predominance. Some series also report a less common older bimodal incidence of the disease. The antibodies target the NC1 domain of the alpha-3 chain of type 4 collagen. The onset may be unprovoked, but there may be a trigger related to infection, smoking, or exposure to inhaled hydrocarbons.
Renal manifestations as the initial clinical finding of anti-GBM disease are most common and are present in 90% of cases; 25% to 60% of patients have both pulmonary and renal findings, and a smaller percentage have solely pulmonary findings. The renal manifestations include hematuria, proteinuria, and casts progressing to renal insufficiency and on biopsy an RPGN/capillaritis with crescent formation. Pulmonary manifestations range from mild hemoptysis to significant alveolar hemorrhage. Imaging may show patchy or diffuse alveolar infiltrates and ground-glass opacities (Figure 1).
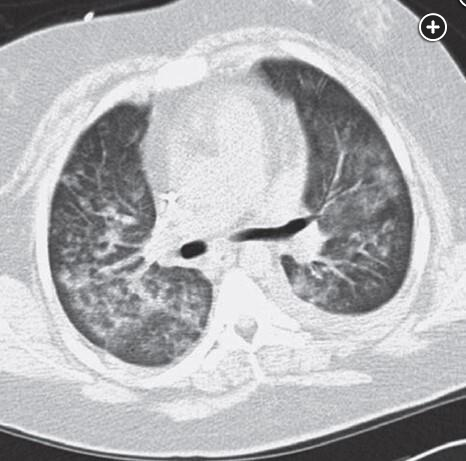
Pulmonary function tests (although rarely performed in the acute setting) may show mild restriction or be normal, and often there is an elevated diffusing capacity due to the presence of blood in the alveoli. If bronchoscopy is performed, the typical findings will be that of DAH manifested by return of progressively bloodier BAL fluid on serial lavage (Figure 2). BAL analysis usually reveals >20% hemosiderin-laden macrophages. The presence of circulating antibodies against the GBM, using a direct enzyme-linked immunoassay, is supportive of the diagnosis, although a renal biopsy is often performed to confirm this.

Treatment of the capillaritis related to anti-GBM disease includes corticosteroids, cyclophosphamide, and plasmapheresis in combination. The benefit of the added plasmapheresis is to remove circulating anti-GBM antibodies. Studies have not shown clear efficacy with rituximab and it is not typically used as a first-line immunosuppressive agent in anti-GBM disease, and plasmapheresis must be part of the initial regimen. No treatment, results in progression of the disease, resulting in chronic renal failure. When treated appropriately, respiratory sequelae are rare, although the renal manifestations can persist.
Corticosteroids alone, which could be considered for idiopathic hemosiderosis, a rare condition of recurrent hemoptysis, anemia, and radiographic findings of DAH in children and young adults, and perhaps vaping-induced lung disease among other conditions, are not appropriate for treating anti-GBM disease. For antineutrophil cytoplasmic antibody (ANCA)-associated vasculitides (not present in this patient), recent studies suggest no additional benefit of plasmapheresis compared with corticosteroids with cyclophosphamide or rituximab, at least for the endpoints of death and end-stage renal disease, and possibly for the DAH component as well. This is based on the PEXIVAS trial where the use of plasma exchange did not reduce the incidence of death or end-stage kidney disease (hazard ratio, 0.86; 95% CI, 0.65-1.13) at 1 year or during the follow-up period of up to 7 years. It has been suggested that plasmapheresis be reserved for severe life-threatening disease in ANCA-related disease, but it is a mainstay in treatment of anti-GBM disease. It should be noted anti-GBM and ANCA vasculitis can occur together.
When anti-GBM disease results in massive hemoptysis, management includes intubation and supportive critical care as indicated. Inhaled tranexamic acid can be used to control significant ongoing bleeding of any etiology, while awaiting more definitive therapy, but is not needed in this case. Bronchial artery embolization by interventional radiology has become a mainstay for focal or localized hemoptysis due to structural abnormalities, such as bronchiectasis, mycetoma, other cavitary disease, or sometimes lung cancer, but would not be indicated in this case because one needs to treat the underlying cause of the DAH. Other causes of DAH include other vasculitides, coagulopathy, bland alveolar hemorrhage, drugs, and inhaled toxins.123456
Links to this note
Footnotes
-
Lazor R, Bigay-Gamé L, Cottin V, et al; Groupe d’Etudes et de Recherche sur les Maladies Orphelines Pulmonaires (GERMOP); Swiss Group for Interstitial and Orphan Lung Diseases (SIOLD). Alveolar hemorrhage in anti-basement membrane antibody disease: a series of 28 cases. Medicine (Baltimore). 2007;86(3):181-193. PubMed ↩
-
Lichtenberger JP 3rd, Digumarthy SR, Abbott GF, et al. Diffuse pulmonary hemorrhage: clues to the diagnosis. Curr Probl Diagn Radiol. 2014;43(3):128-139. PubMed ↩
-
McAdoo SP, Pusey CD. Antiglomerular basement membrane disease. Semin Respir Crit Care Med. 2018;39(4):494-503. PubMed ↩
-
Sanders JS, Rutgers A, Stegeman CA, et al. Pulmonary: renal syndrome with a focus on anti-GBM disease. Semin Respir Crit Care Med. 2011;32(3):328-334. PubMed ↩
-
Walsh M, Merkel PA, Peh CA, et al; PEXIVAS Investigators. Plasma exchange and glucocorticoids in severe ANCA-associated vasculitis. N Engl J Med. 2020;382(7):622-631. PubMed ↩