subdural hematoma management
- related: Neurology
- tags: #literature #neuro
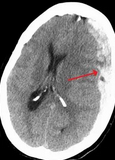
Subdural hematoma is a common presentation for elderly patients who have a fall. Surgical or medical interventions are based on the patient’s physical examination results, regardless of the size of the subdural collection or edema. Because this patient is clinically awake and oriented and the hemiparesis is mild, the correct approach is to delay treatment of the subdural hematoma until the patient develops symptoms or has evidence of recurrence.
The treatment for subdural hematoma consists of graded treatments based on clinical examination results during the course of the injury. Conservative treatment is favored for patients with normal or near-normal examination results such as this one. In patients with impaired consciousness, signs of uncus herniation, or significant focal neurological deficits, surgical evacuation is the treatment of choice. Initially, surgery entails a large craniotomy to remove the clotted hemorrhage. Over days to weeks, the hemorrhage begins to liquefy, which allows for more conservative surgical approaches such as burr hole evacuation or a subdural evacuation port system drain procedure to drain the collection slowly. Trying to get the patient to the point where the hemorrhage has liquefied before attempting surgery decreases morbidity. In patients who have recurrent hemorrhage or hemorrhage expansion, embolization of the middle meningeal artery via an endovascular approach has been shown to prevent further recurrence of hemorrhage. This patient has had only one hemorrhage and no expansion and so would not be a candidate for this approach. Prophylactic intubation and mechanical ventilation are not warranted in this patient who has no respiratory distress and for whom emergency surgery is not planned.12