thromboelastogram TEG
- related: Hematology
- tags: #literature #hemeonc
Thromboelastography (TEG) or the similar rotational thromboelastometry are functional viscoelastic hemostatic assays that can identify fibrinolysis and hence guide the clinician to employ agents such as tranexamic acid, an inhibitor of fibrinolysis, to treat this contribution to hemorrhage (choice C is correct). In the performance of TEG, a small sample of blood is taken and placed in a device that rotates slowly, simulating venous stasis, and the clot formed can be characterized in a number of ways, typically by computer analysis of the dimensional aspects of the formed clot. This technology is readily available for use at the bedside; is readily available as a point-of-care test; and has been adopted in many trauma, liver transplant, and postoperative ICUs. An example of reported measurements is shown (Figure 1).
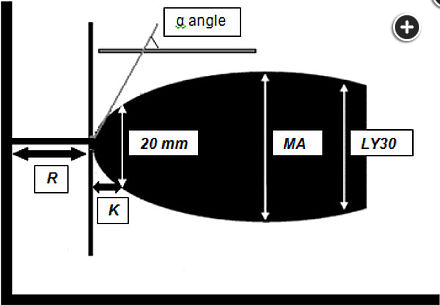
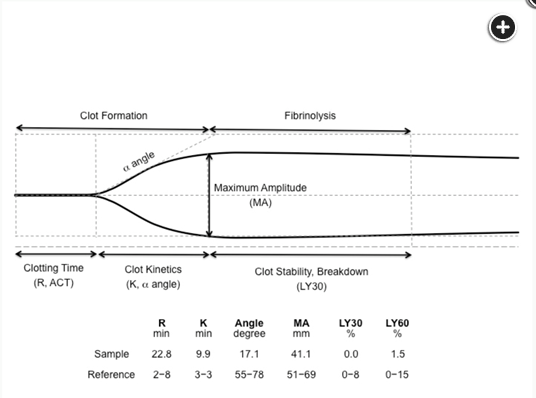
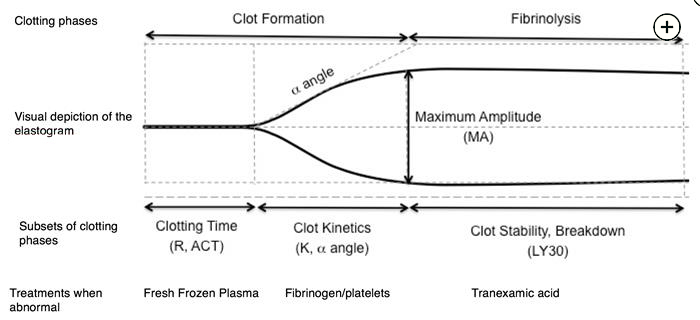
The angle of deflection in the initial clot firmness curve reflects the slope in the clotting curve and is an indicator of the velocity of initial clot amplification. The maximal amplitude of clot firmness indicates the overall clot strength. Abbreviations: R, reaction time; ACT, activated clotting time; K, coagulation time; LY, clot lysis.1
- The R (Reaction) value represents the first time of recognition of clot formation
- the K (Kinetic) value, the time from onset of clot formation until formation of a 20-mm clot
- -angle measures rate of rise of clot strength
- the MA (Maximum Amplitude) value, a measure of the strength of the clot
- LY30 value, the percentage of clot lysis observed at 30 min.234
- R: give clotting factors such as 4f pcc
- both K-time and -angle are affected by fibrinogen: give cryoprecipitate
- MA depends on platelet number and function
- increased by desmopressin and plt transfusion
- decreased by thrombocytopenia and impaired plt function
- LY30: give antifibrinolytic therapy such as TXA or aminocaproic acid
In the CRASH-2 trial, tranexamic acid reduced bleeding and mortality after major trauma.
The utility of TEG has been evaluated in subsets of patients at risk of major bleeding. In a randomized prospective trial in patients with advanced liver disease who were undergoing invasive procedures, TEG-guided transfusion resulted in significantly lower transfusions of blood products without a concomitant increase in bleeding complications. In a separate randomized controlled trial, patients with cirrhosis stratified to a TEG-guided transfusion strategy received significantly fewer blood products and had fewer transfusion-related adverse events than did patients randomly assigned to a standard coagulation-testing-based approach. Similar observations have been made in patients undergoing liver transplant. TEG has also been shown to reduce unnecessary blood transfusions in patients undergoing cardiac surgery and is now recommended by the UK National Institute for Health and Clinical Excellence to monitor hemostasis in patients undergoing cardiac surgery and by the European Society of Anesthesiology to guide transfusion practices in patients with perioperative or peripartum bleeding.
Clot stability and breakdown are measured by the percentage decrease in clot stability 30 and 60 min after MA (LY30, LY60), which provides information on the degree of fibrinolysis. An increased LY30, as in this case, might in general prompt consideration of treatment with tranexamic acid (TXA), but this is not an option in the question. TXA has been demonstrated in the CRASH-2 trial to decrease all-cause mortality among patients presenting with or at risk for traumatic hemorrhage, when TXA is administered within the first 8 h (especially the first 3 h) of presentation. On the basis of current evidence, the patient would thus be receiving TXA regardless of the TEG results. The TEG in this case would not suggest administration of either fresh frozen plasma or cryoprecipitate, as the normal K time and normal α angle argue against infusion of coagulation factors and fibrinogen (choices B and C are incorrect).
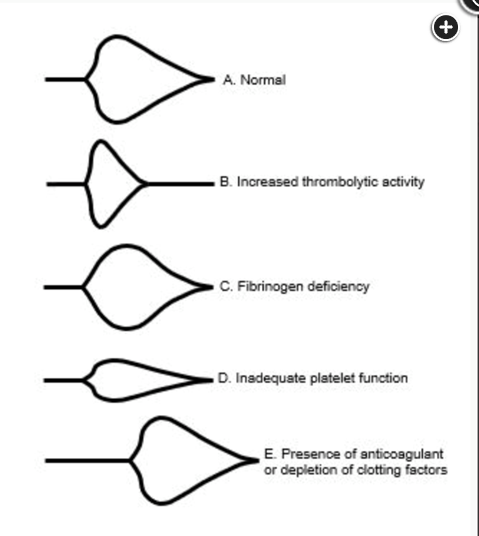
Pattern recognition is a good introduction to the use of TEG. Figure 3 shows graphic representations of characteristic TEG patterns that are commonly encountered in patients with coagulopathy. Panel A is the normal control. Panel B demonstrates increased fibrinolytic activity (LY-30). Panel C shows an increased K-time and reduced α-angle characteristic of impaired fibrinogen activity, and the reduced MA in Panel D reflects reduced maximal clot strength from impaired platelet function or number. Finally, the prolonged R-time in Panel E is seen in patients with deficiencies in clotting factors and treated with anticoagulants.
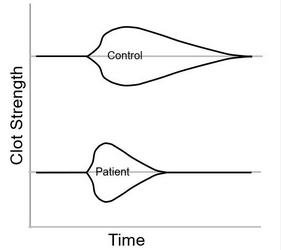
In the case presented in the question, the R-time, K-time, α-angle, and MA are similar to the control values. However, the LY-30 is significantly elevated (ie, clot lysis is accelerated). In a patient who is bleeding, administration of an antifibrinolytic, like tranexamic acid, would be indicated (choice C is correct). Additional platelet transfusions would be indicated only if the maximal viscoelastic amplitude of the clot was reduced (choice A is incorrect). Cryoprecipitate is enriched with fibrinogen. This patient’s normal K-time and α-angle suggest that cryoprecipitate would not be useful to stanch the bleeding (choice B is incorrect). 4-Factor prothrombin complex concentrate is made from the supernatant of cryoprecipitate. It is enriched with clotting factors II, VII, IX, and X and is most useful for reversing warfarin anticoagulation. This patient’s normal R-time suggests that there is not a significant warfarin effect contributing to this patient’s bleeding (choice D is incorrect).56789
Links to this note
Footnotes
-
da Luz LT, Nascimento B, Rizoli S. Thrombelastography (TEG®): practical considerations on its clinical use in trauma resuscitation. Scand J Trauma Resusc Emerg Med. 2013;21:29. PubMed ↩
-
Sarode K, Hussain SS, Tyroch A, et al. A review of the current role of blood clotting analyzers in clinical practice. Cardiovasc Hematol Disord Drug Targets. 2017;17(3):167-179. PubMed ↩
-
Sharp G, Young CJ. Point-of-care viscoelastic assay devices (rotational thromboelastometry and thromboelastography): a primer for surgeons. ANZ J Surg. 2019;89(4):291-295. PubMed ↩
-
Cannon JW, Dias JD, Kumar MA, et al. Use of thromboelastography in the evaluation and management of patients with traumatic brain injury: a systematic review and meta-analysis. Crit Care Explor. 2021;3(9):e0526. PubMed ↩
-
Drumheller BC, Stein DM, Moore LJ, et al. Thromboelastography and rotational thromboelastometry for the surgical intensivist: a narrative review. J Trauma Acute Care Surg. 2019;86(4):710-721. PubMed ↩
-
Kim Y, Patel SS, McElroy IE, et al. A systematic review of thromboelastography utilization in vascular and endovascular surgery. J Vasc Surg. 2022;75(3):1107-1115. PubMed ↩
-
CRASH-2 trial collaborators; Shakur H, Roberts I, Bautista R, et al. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): a randomised, placebo-controlled trial. Lancet. 2010;376(9734):23-32. PubMed ↩
-
Subramanian M, Kaplan LJ, Cannon JW. Thromboelastography-guided resuscitation of the trauma patient [published online ahead of print October 9, 2019]. JAMA Surg. doi: 10.1001/jamasurg.3136. PubMed ↩